In recent years, SO4•- advanced oxidation processes (SR-AOPS) has become a research hotspot due to its wide application range and strong oxidation capacity. Among the various methods of activating PMS to produce SO4•-, the transition metal activation reaction system tends to show higher catalytic performance and lower energy requirements, thus it is an important SO4•- activation technique. However, the single transition metal catalyst and multi-supported catalyst reported in previous studies often have problems such as high metal ion dissolution and poor catalyst stability in the activated PMS system. In addition, when heterogeneous nanocatalysis materials are used in the suspension system, it is difficult to recover catalyst and easy to agglomerate, which is not conducive to the exposure of active sites.
Recently, Professor Cheng Xiuwen's research group in our college has made a series of research progress in exploring the construction strategy of efficient and stable heterogeneous PMS catalytic reaction system.
Based on relevant theoretical analysis, the research group determined that LDHs and PBAs have good stability due to their integrity and internal solid skeleton, and have good catalytic performance due to their internal combination of multiple types and multi-valence transition metals. Based on this, researchers constructed the PMS catalytic reaction system based on LDHs and PBAs respectively, optimized the catalyst composition and the synthesis process parameters to improve its performance and further promote the efficient and rapid degradation of pollutants in water. Then, based on the antibiotics degradation experiment and reaction kinetics study, researchers explained the influence of various parameters on the reaction system. In particular, by exploring the types of active oxidation molecules generated in the LDHs/PMS reaction system, the mechanism of the pollutant degradation process is clarified, and the mechanism of action of the system in the efficient treatment of emerging micro-pollutants is revealed (Figure 1), which provides a theoretical basis for the research and development of efficient water purification technology and environmental functional nanomaterials.
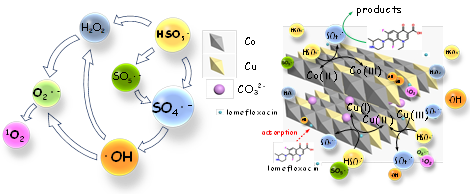
Figure 1 Mechanism of free radical transformation path and pollutant degradation in LDHs/PMS reaction system
Photocatalytic technology is an environment-friendly pollution control technology, which has attracted much attention due to its advantages of strong oxidation ability and non-selectivity of •OH generated by light induction. Photoactivation is a typical way of persulfate activation in higher oxidation systems. Based on the above ideas and combined with the previous research results, the research group constructed a variety of visible light assisted heterogeneous catalysts to activate PMS systems (Figure 2), so as to achieve rapid and efficient degradation of micro-pollutants in the water while reducing the energy consumption of the reaction system.

Figure 2 Mechanism of visible light assisted heterogeneous PMS activation reaction based on (a) MnO2/Fe2O3 nano-mixture and (b) CuFe PBAs derivatives
In addition, the research group also designed and developed an economical and effective composite catalytic membrane, with a view to effectively activate PMS to treat hospital wastewater and solve the problem of particle agglomeration and recovery. For this, The nanoparticles with high catalytic performance were added into the polymer film matrix by the deposition method (Figure 3). Then, by optimizing its preparation technology parameters, constructing a reaction system for activating PMS to treat hospital medical wastewater with a composite catalytic membrane, exploring its PMS activation performance and reusability of the reaction system, the new research ideas for improving the practical application potential of environmental functional nanomaterials were provided.
The above findings have recently been published in Applied Catalysis B: Environmental, Chemical Engineering Journal and Journal of Hazardous Materials (These publications are all in the first district of the Chinese Academy of Sciences, with impact factors of 16.683, 10.652 and 9.038 respectively), the first author of the paper is Guo Ruonan, a master's degree candidate in environmental engineering of class 2018 (graduated one year ahead of schedule and is pursuing a doctorate at Tsinghua University), and the College of Earth and Environmental Sciences of Lanzhou University is the first signature institution. The research was supported by general projects of the National Natural Science Foundation of China.

Figure 3 SEM images of (a) CGF, (b) LDH(6.25%)@CGF, (c) PBA(9.09%)@CGF, (d) PAN, (e) LDH(9.09%)@PAN and (f) PBA(6.25%)@PAN
Links to papers:
https://www.sciencedirect.com/science/article/abs/pii/S0926337320307128
(Sulfamethoxazole degradation by visible light assisted peroxymonosulfate process based on nanohybrid manganese dioxide incorporating ferric oxide, Applied Catalysis B: Environmental,(2020) 119297.)
https://www.sciencedirect.com/science/article/abs/pii/S1385894720318040
(Efficient degradation of sulfamethoxazole by CuCo LDH and LDH@fibers composite membrane activating peroxymonosulfate, Chemical Engineering Journal, 398 (2020) 125676.)
https://www.sciencedirect.com/science/article/abs/pii/S1385894720316843
(In situ preparation of carbon-based Cu-Fe oxide nanoparticles from CuFe Prussian blue analogues for the photo-assisted heterogeneous peroxymonosulfate activation process to remove lomefloxacin, Chemical Engineering Journal, 398 (2020) 125556.)
https://www.sciencedirect.com/science/article/abs/pii/S0304389420309559
(Efficient degradation of lomefloxacin by a Co-Cu-LDH-activating peroxymonosulfate process: Optimization, dynamics, degradation pathway and mechanism, Journal of Hazardous Material, (2020) 122966.)